 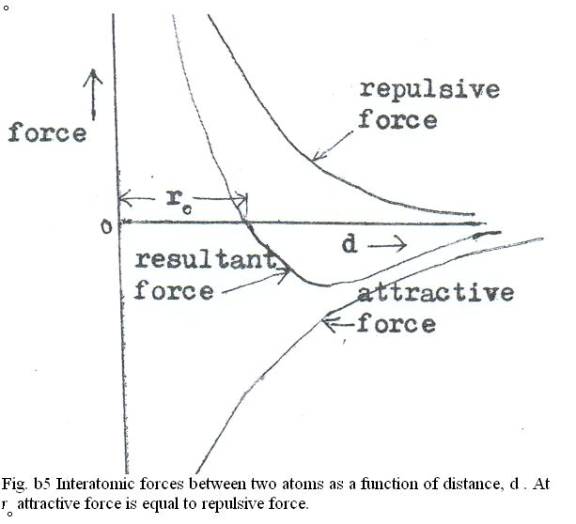
electronegativity: The tendency of an atom to attract electrons to itself.electrolyte: An ionic compound which dissolves in H2O, making the resulting solution capable of conducting electricity.Ionic solids form crystalline lattices, or repeating patterns of atoms, with high melting points, and are typically soluble in water.They both achieve a more stable electronic configuration through this exchange. A cation is formed when a metal ion loses a valence electron while an anion is formed when a non-metal gains a valence electron.Ionic bonds are formed between cations and anions.The Editors of Encyclopaedia Britannica This article was most recently revised and updated by Erik Gregersen. 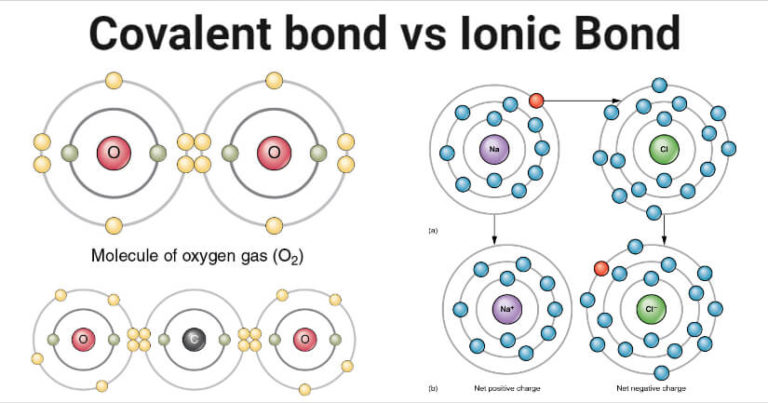
Hydrogen bonds between nitrogenous bases in nucleotides on the two strands of DNA ( guanine pairs with cytosine, adenine with thymine) give rise to the double-helix structure that is crucial to the transmission of genetic information. Hydrogen bonding between amino acids in a linear protein molecule determines the way it folds up into its functional configuration. Water is also a good solvent for ionic compounds and many others because it readily forms hydrogen bonds with the solute. Because of its extensive hydrogen bonding, water (H 2O) is liquid over a far greater range of temperatures that would be expected for a molecule of its size. Mainly through electrostatic attraction, the donor atom effectively shares its hydrogen with the acceptor atom, forming a bond. 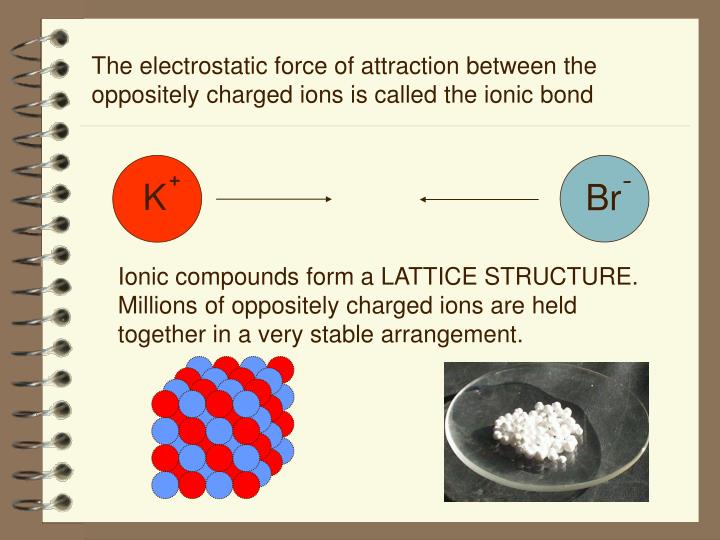
The other atom of the pair, also typically F, N, or O, has an unshared electron pair, which gives it a slight negative charge. One atom of the pair (the donor), generally a fluorine, nitrogen, or oxygen atom, is covalently bonded to a hydrogen atom (―FH, ―NH, or ―OH), whose electrons it shares unequally its high electron affinity causes the hydrogen to take on a slight positive charge. Hydrogen bonds can exist between atoms in different molecules or in parts of the same molecule. Hydrogen bonding, interaction involving a hydrogen atom located between a pair of other atoms having a high affinity for electrons such a bond is weaker than an ionic bond or covalent bond but stronger than van der Waals forces. SpaceNext50 Britannica presents SpaceNext50, From the race to the Moon to space stewardship, we explore a wide range of subjects that feed our curiosity about space!.Learn about the major environmental problems facing our planet and what can be done about them! Saving Earth Britannica Presents Earth’s To-Do List for the 21st Century.100 Women Britannica celebrates the centennial of the Nineteenth Amendment, highlighting suffragists and history-making politicians.COVID-19 Portal While this global health crisis continues to evolve, it can be useful to look to past pandemics to better understand how to respond today.Student Portal Britannica is the ultimate student resource for key school subjects like history, government, literature, and more.This Time in History In these videos, find out what happened this month (or any month!) in history.#WTFact Videos In #WTFact Britannica shares some of the most bizarre facts we can find.Demystified Videos In Demystified, Britannica has all the answers to your burning questions.Britannica Classics Check out these retro videos from Encyclopedia Britannica’s archives. 
Britannica Explains In these videos, Britannica explains a variety of topics and answers frequently asked questions. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
